|
The fundamental premise of this theory can be stated as follows:Įlectrons in bonded atoms occupy spatially oriented orbitals in such a way as to minimize electron-electron repulsions arising mainly from electrostatic (coulombic) forces. 
This approach, which was originated by Nyholm and Gillespie in the 1950's, has become known as the Valence Shell Electron Pair Repulsion Theory, or VSEPR. One of the most successful approaches to predicting the shapes of molecules is based almost solely on considerations of how best to minimize electron-pair repulsions about a central atom. Knowing the shape of a molecule enables us to predict whether or not it has an electrical polarity, which is an important property determining how the species interacts with other molecules. This method is called the Valence Shell Electron Repulsion Theory, or VSEPR for short. Although the Lewis structures themselves do not convey shape information, they can be used as the starting point for applying a conceptually simple but powerful approach to predicting molecular geometries. These molecular shapes are very important to understanding how molecules interact with each other, both chemically and physically. Know how bond polarity and molecular shape combine to make a molecule polar or nonpolar.Ĭontrary to the impression that Lewis structures may give, many molecules have three-dimensional geometries.Understand how bond pairs and lone pairs about a central atom interact to produce the molecular shape. 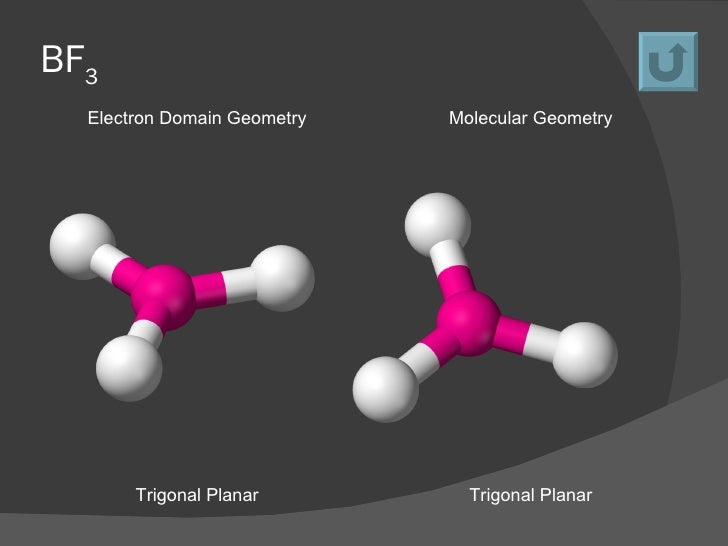
Know the expected geometries for one through six electron domains about a central atom.Understand the basis of the VSEPR theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
